Let us inaugurate this blogging adventure with some of my favorite primates. Tarsiers’ unique, derived morphology, possible relationships to anthropoids and the fossil omomyids, and and apparently deep history as an independent lineage make them particularly intriguing from the perspective of primate paleontology.
View from the Holocene
Tarsiers are famously weird. They appear to be the only totally faunivorous group of primates, eating a diet of exclusively arthropods and small vertebrates. They possess the largest eyes relative to body size of any mammal and are entirely nocturnal. Tarsiers are adapted to bounding long distances between vertical supports in a system of locomotion known as vertical clinging and leaping, of which tarsiers constitute the paradigmatic example (Napier and Walker 1967). This locomotor mode is present to varying degrees in many primates, but none have committed to it as definitively as these Southeast Asian prosimians. Tarsiers draw their name from their elongated tarsal bones, which are an adaptation to this peculiar lifestyle and give extra power to their leaps. They have also fused their tibia and fibula into a single bone, an adaptation found in other highly saltatory taxa such as frogs.
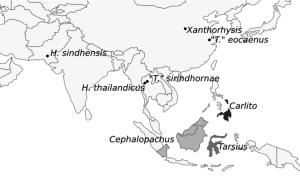
The taxonomy of living tarsiers was substantially revised in 2010 by Groves and Shekelle, who arranged crown tarsiers into the three genera Cephalopachus, Carlito, and Tarsius. Cephalopachus contains the single species C. bancanus, the Western tarsier, indigenous to parts of the Pleistocene Sunda Shelf in western Indonesia (Borneo, southern Sumatra, and a few smaller islands). Carlito contains the species Ca. syrichta, the Philippine tarsier, and is endemic to islands of the southern Philippines that would have formed the Ice Age landmass Greater Mindanao. Both of these genera may contain more cryptic species yet to be described, as the variability within each of their monotypic species has yet to be fully studied. Tarsius, which formerly included all living tarsiers, was restricted by Groves and Shekelle (2010) to a complex of at least 8 species, more possibly still undescribed, from the Indonesian island of Sulawesi, collectively referred to as the Eastern tarsier. These formerly cryptic species have been postulated on the basis of genetic as well as acoustic and morphological data, and likely reflect the complex geological history of Sulawesi, which coalesced relatively recently from the collision of multiple smaller islands, including a portion of the former Sundaland (Merker et al. 2009).
The mountain tarsier, T. pumilus, is one species from Sulawesi that deserves particular mention. Mountain tarsiers are smaller bodied than any other living tarsier, and differ in a suite of morphological and acoustic characters (Musser and Dagosto 1987). They are also the only tarsiers to occur at elevations above 1500 m, inhabiting the montane regions of central Sulawesi. T. pumilus is known from only three museum specimens, none of them suitable for genetic analysis, and has never been observed alive. Shekelle (2008b) proposed that its small size, apparent lack of duet calling, and fully furred tail indicate that the mountain tarsier may be the most primitive of all living tarsiers, and possibly sister to the entire crown radiation*. Without further specimens of this species this hypothesis is difficult to test, however.
The three genera of living tarsiers represent a morphological cline in most of the unique tarsier adaptations, with Cephalopachus generally representing the most extremely elaborated morphology, Tarsius the most restrained, and Carlito somewhere intermediate (Shekelle 2008a). This is reflected in eye size (largest in Cephalopachus, smallest in Tarsius), limb elongation (highest in Cephalopachus), dental size (largest in Cephalopachus), etc. This cline is also reflected in the degree of vertical clinging and leaping practiced by members of each genus, with Cephalopachus the most specialized (Crompton et al. 2010). There are also differences in sociality among tarsier genera, with Cephalopachus the most solitary in habits and at least some species of Tarsius known to be fairly gregarious, sharing sleeping nests and foraging in family groups (Gursky 2003).
The lack of a clear outgroup makes it difficult to establish character polarity for these traits, so any of the three taxa may well exhibit the more plesiomorphic state for the group. T. pumilus also complicates this picture, as it sometimes approaches Cephalopachus in its trait elaboration relative to body size (in eye diameter for example), but it is unclear if this is a reflection of dwarfing or of a plesiomorphic retention. The rarity of T. pumilus has also excluded it from all molecular studies of tarsier phylogeny, further mystifying its position relative to the rest of the Tarsius species complex and the remainder of the tarsiiform crown radiation.
The introduction of molecular phylogenetic approaches to tarsier evolution have yielded interesting results with respect to the age of diversification of the living members of the former genus Tarsius. The latest study to examine the issue (Springer et al. 2012) reported a middle Miocene (18.64 Ma) mean date of divergence among all living tarsiers, more distant than the mean divergence date among living hominoids (17.36 Ma). The antiquity of living tarsiids would seem to support the proposed erection of three genera for the Western, Philippine, and Eastern tarsiers.
The Fossils

Hypothetical relationships between living and fossil tarsiers, drawing on Beard (1998) and Zijlstra (2013).
It should first be mentioned that Tarsiiformes are now often thought to include the large radiation of mostly Eocene primates known as the omomyids, as well as potentially even more primitive taxa such as the newly described Archicebus achilles from the earliest Eocene of China (Ni et al. 2013), which together with anthropoids form the Haplorhini. The internal relationships among haplorhines are somewhat controversial, with sister group relationships between tarsiers and omomyids, tarsiers and anthropoids, or anthropoids and omomyids all proposed, as well as the origins of either tarsiers or anthropoids within a paraphyletic omomyidae. I won’t specifically deal with this issue in this post, except to note that the rather great antiquity of tarsiids (45 Ma, the middle Eocene), implies that any origin of tarsiers within omomyids must have occurred very early in the evolution of the radiation. Six fossil tarsier species are known. Upon description three were placed in what was then the crown genus Tarsius. The restriction of Tarsius to the Sulawesi species group makes the assignment of such ancient and likely stem fossil taxa to this genus dubious, however. I’ve followed the genus assignments of Zijlstra et al. (2013) below.
Xanthoryhisis tabrumi was described from a complete left dentary from the middle Eocene of Shanxi Province, China (Beard 1998). X. tabrumi shares with modern tarsiers distinctive sharp crests emerging from the protoconids of both premolars and giving them a somewhat pyramidal shape. Xanthorhysis also appeared to possess a relatively large canine, as in tarsiers and differing from the character state in omomyids, although this may reflect the primitive state in primates (it is present in Archicebus, for example [Ni et al. 2013]) and therefore not be a synapomorphy of tarsiers. In molar morphology, Xanthorphysis and modern tarsiers share features such a metaconid widely spaced from both the protoconid and paraconid and strongly developed entoconids with sharp cresting.
There are several differences in molar morphology between X. tabrumi and modern tarsiers, however, some of which are traits shared between Xanthorhysis and omomyids and may represent the ancestral state in tarsiiformes. These include a relatively long, narrow, and low crowned molar profile. Beard’s (1998) parsimony analysis incorporating the Western, Eastern, Philippine, and mountain tarsiers as well as the omomyid species Teilhardina belgica, Steinius verspertinus, and Shoshonious cooperi and the adapoid Cantius supports the placement of Xanthorhysis as sister to the remainder of the tarsiers.
“Tarsius” eocaenus was originally described based on three isolated teeth (Beard et al. 1994). It is now also known from a partial upper maxilla from which some details of the facial anatomy could be reconstructed (Rossie et al 2006). Beard et al. (1994) placed “T.“ eocaenus in Tarsius, holding that its dental anatomy was insufficiently distinguishable from the modern taxa. At around 45 million years old, however, this would constitute an extraordinary temporal extension for Tarsius, even before the revision of the modern species into three genera. This led Simons (2003) and Zijlstra et al (2013) to advocate placing “T.” eocaenus in a new genus, although this has yet to be undertaken.

Scaled molar dentitions from Beard (1998) of fossil tarsiers. B, Xanthorhysis tabrumi; C, Tarsius bancanus; D, “Tarsius” eocaenus; E, Hesperotarsius thailandicus.
The principal dental difference identified by Beard et al. (1994) between “T.” eocaenus and the modern taxa was the extremely small size of its dentition, smaller even than T. pumilus. Beard et al. (1998) also noted somewhat lower crown height in “T.” eocaenus, which appears to be the primitive state for tarsiers and is shared with X. tabrumi.
The facial fragment described by Rossie et al. (2006) preserves a complete P3 and partial alveoli of surrounding teeth as well as internal sinus and foraminal anatomy and a small portion of the orbital rim, all of which are described as virtually identical to modern tarsiers. The bell-shaped profile of the dental arcade is also strikingly similar to the modern condition. The infraorbital foramen appears to be relatively small in “T.” eocaenus, also similar to the state in modern tarsiers and that in anthropoids, but potentially different from that in omomyids. This foramen is associated with facial vibrissae and is smaller in the dry-nosed haplorhine primates than in the wet-nosed strepsirrhine primates. This may indicate that tarsiers had evolved an anatomically haplorhine nose by the middle Eocene.
“Tarsius“ sirindhornae was described on the basis of a large collection of cranial and dental material from the middle Miocene of Thailand (Chaimanee et al. 2010). It is the best represented fossil tarsiid. It is dated to around 13.3-13.1 Ma. Like “T.” eocaenus, its membership in the restricted genus Tarsius would be unlikely, although its age after the ~18 Ma divergence between Eastern and Western tarsiers makes it possible that it may represent a member of crown group tarsiidae.
“T.” sirindhornae appears to be the largest fossil tarsier species, at an estimated 90-180 g, perhaps as large or larger than Cephalopachus bancanus. Its teeth also show a relatively low cusped profile and high degree of wear, unusual for a tarsiid, and perhaps representing a less faunivorous diet. Chaimanee et al. (2010) compare this to the wear observed in the molars of T. pumilus by Musser and Dagosto (1987) which is similarly mysterious as virtually nothing is known of mountain tarsier behavior, but may also reflect a non-faunivorous, or at least more abrasive, dietary component. It is curious to see such a similar adaptation in what are among the largest and smallest known tarsiers.
The relatively widely spaced premolar roots and long tooth rows of “T.” sirindhornae indicate that it probably had a longer snout than any living tarsiers. However, morphometric reconstruction indicate that it possessed the fully enlarged orbits typical of modern tarsiers, dating this trait at least to the middle Miocene.
Hesperotarsius thailandicus was originally described as Tarsius thailandica (revised to Tarsius thailandicus for grammatical reasons) from a set of isolated teeth from the middle Miocene of Thailand (Ginsburg and Mein 1987). It appears to be roughly contemporaneous with “T.” sirindhornae. It, like the Eocene taxa, appears to closely resemble living tarsiers in general morphology. In particular, its closely spaced premolar roots indicate that it had a shorter snout than “T.“ sirindhornae (Chaimanee et al. 2010). However, its close resemblance in molar morphology to Hesperotarsius sindhensis led to its tentative assignment to that genus by Zijlstra et al. (2013).
Hesperotarsius sindhensis is the furthest afield of all of the certain fossil tarsiids. It was described from dental material from the Miocene of southern Pakistan (Zijlstra et al. 2013). This is not as shocking as might first appear, as other Southeast Asian forest taxa such as treeshrews have been described from the same deposits, but it does represent a significant geographic range extension for the lineage. H. sindhensis is dated at between 18-16 Ma, somewhat older than the Thai Miocene species.
H. sindhensis shares with all fossil tarsiers relatively elongate and low molars compared with the modern taxa, which appear to possess a relatively derived condition in this regard. However, the molar narrowing and elongation in H. sindhensis is extreme, similar to X. tabrumi.
H. sindensis is perhaps most interesting for its biogeographic implications. Southern Pakistan in the Miocene appeared to possess a relatively stable, warm climate, similar to that of Southeast Asia today, and in contrast with the more seasonal climates known from northern Pakistan (the famous Siwalik deposits) and southern China, which lack fossil tarsiers in the Miocene. This suggests that tarsiers once inhabited a large range of stable tropical forest in Asia that today persists only in the regions of Southeast Asia to which they are currently restricted.
Summary
A few interesting conclusions might be drawn from what is known of the tarsier fossil record. Tarsiers appear to have been remarkably morphologically stable for at least the last 45 million years. This has important implications for our own anthropoid lineage, as most workers currently consider tarsiers to the be the sister group to anthropoids. Molecular phylogenetics date the divergence between tarsiers and anthropoids at 61.16 Ma (Springer et al. 2013), relatively close to the Eocene date of the earliest tarsiers. This may indicate that our last common ancestor with tarsiers was relatively tarsier-like itself in some respects, a result supported by the morphology of the 55 Ma probable early haplorhine Archicebus achilles (Ni et al. 2013). It was probably unlike tarsiers in possessing enlarged orbital morphology, however, as this appears to be a secondary adaptation to nocturnality in an animal that has lost the tapetum lucidum of most nocturnal mammals and gained a fovea useful in diurnal predation (Chaimanee et al. 2010). This suggests that the LCA of anthropoids and tarsiers may have been diurnal, an interpretation also supported by the apparently diurnal adaptation of Archicebus (Ni et al. 2013).
The remarkable stability of tarsier adaptation may reflect the stability of their preferred habitat. Forests similar to those present in island Southeast Asia once covered much of the Asian continent. As these forests contracted, the range of tarsiers appeared to contract as well. They now remain in what are probably the oldest forests on earth, structurally unchanged since the Eocene (Jablonski 2003).
Because no fossil tarsiids are known from within the current distribution of tarsiers, and none are clearly attributable to any living genus, the ability of the fossil record to clarify the historical biogeography of living tarsiers is limited. However, it does appear clear that tarsiers originated on the landmass of Asia, which was once attached to the current range of the Western Tarsier as part of Sundaland. It therefore appears likely that tarsiers first dispersed from Sundaland to the proto-Sulawesi archipelago, perhaps carried on a moving terrain as that island began to coalesce in the Miocene, and then from Sundaland to Greater Mindanao in the Philippines some millions of years later, as hypothesized by Shekelle (2008a). Different Sulawesi species developed on different islands of the proto-sulawesi group and then were brought into parapatric contact by the formation of the island. This would account for the speciosity of the Sulawesi group and the apparently deep divergence between these species and the clade of Philippine and Western tarsiers.
All of these conclusions are built upon the understanding that afrotarsiids are not closely related to tarsiids and therefore not directly part of this story. Part II will examine that enigmatic group and its possible history with tarsiids.
*A crown group is a clade of all living members of a taxon and those fossil species more closely related to one of the living members than to any of the others. This is opposed to a stem group, which is more closely related to the living taxa as a group than to any other living group, but is outside of the clade containing all of the living members exclusively. All living birds, for example, are crown birds, as would be any fossil bird more closely related to a sparrow, say, than an ostrich. Any fossil species outside of the living bird clade, but more closely related to birds as a group than to crocodylians would be a stem bird. “Stem bird” is therefore an acceptable substitute for “non-Avian dinosaur,” if you are so inclined.
References
K. C. 1998. A new genus of tarsiidae (Mammalia: Primates) from the middle Eocene of Shanxi Province, China, with notes on the historical biogeography of tarsiers. Bull. Carnegie Mus. Nat. Hist 34:260–277.
Beard, K. C., T. Qi, M. R. Dawson, B. Wang and C. Li. 1994. A diverse new primate fauna from middle Eocene fissure fillings in southeastern China. Nature 368:604–609.
Chaimanee, Y., R. Lebrun, C. Yamee and J.-J. Jaeger. 2010. A new Middle Miocene tarsier from Thailand and the reconstruction of its orbital morphology using a geometric − morphometric method. Proceedings of the Royal Society B 278:1956–1963.
Crompton, R. H., M. L. Blanchard, S. Coward, R. M. Alexander and S. K. Thorpe. 2010. Vertical Clinging and Leaping Revisited: Locomotion and Habitat Use in the Western Tarsier, Tarsius bancanus Explored Via Loglinear Modeling. International Journal of Primatology 31:958–979.
Ginsburg, L. and Mein, P. 1987. Tarsius thailandica nov. sp., preimier Tarsiidae (Primates, Mammalia) fossile d’Asie. C R Acad Sci II, 304:1213-1215.
Groves, C. and M. Shekelle. 2010. The genera and species of Tarsiidae. International Journal of Primatology 31:1071–1082.
Gursky, S. 2003. The behavioral ecology of the spectral tarsier, Tarsius spectrum. Evolutionary Anthropology 11:226–234.
Jablonski, N. 2003. The evolution of the tarsiid niche. Pp. 35-49 in Tarsiers: Past, present, and future. Rutgers UP, Rutgers.
Merker, S., C. Driller, D. Perwitasari-Farajallah, J. Pamungkas and H. Zischler. 2009. Elucidating geological and biological processes underlying the diversification of Sulawesi tarsiers. PNAS 106:8459–8464.
Musser, G. G. and M. Dagosto. 1987. The Identity of Tarsius pumilus, a pygmy species endemic to the montane mossy forests of central Sulawesi. American Museum Novitates 2867:1–53.
Napier, J. R. and Walker, A. C. 1967. Vertical clinging and leaping – A newly recognized category of locomotor category of primates. Folia Primatologica 6:204-219.
Ni, X., D. L. Gebo, M. Dagosto, J. Meng, P. Tafforeau, J. J. Flynn, et al. 2013. The oldest known primate skeleton and early haplorhine evolution. Nature 498:60–4.
Rossie, J. B., X. Ni and K. C. Beard. 2006. Cranial remains of an Eocene tarsier. PNAS 103:4381–5.
Shekelle, M. 208a. The history and mystery of the mountain tarsier, Tarsius pumilus. Primate Conservation 23:121–124.
Shekelle, M. 2008b. Distribution and biogeography of tarsiers. Pp. 13–27 in Primates of the Oriental Night. LIPI Press, Bogor.
Simons, E. L. 2003. The fossil record of tarsier evolution. Pp. 9-34 in Tarsiers: Past, present, and future, Rutgers UP, Rutgers.
Springer, M. S., R. W. Meredith, J. Gatesy, C. a Emerling, J. Park, D. L. Rabosky, et al. 2012. Macroevolutionary dynamics and historical biogeography of primate diversification inferred from a species supermatrix. PloS one 7:e49521.
Zijlstra, J. S., L. J. Flynn and W. Wessels. 2013. The westernmost tarsier: a new genus and species from the Miocene of Pakistan. Journal of human evolution 65:544–50.







Pingback: Tarsiers (?) in Time, Part II | Of Omomys and Men